Zeroth Law of Thermodynamics
Visualizing thermal equilibrium between three systems as per textbook illustration
Tip: Follow the steps to visualize the Zeroth Law of Thermodynamics
The Zeroth Law of Thermodynamics
"Two systems in thermal equilibrium with a third system separately are in thermal equilibrium with each other."
1 Initial Setup
Systems A and B are separated by an adiabatic wall, while each is in contact with system C via conducting walls.
2 Thermal Equilibrium with C
Systems A and B come to thermal equilibrium with system C. Their temperatures become equal to C's temperature.
3 Test the Zeroth Law
When the adiabatic wall between A and B is replaced by a conducting wall, they are found to be in thermal equilibrium with each other.
Ready to begin simulation
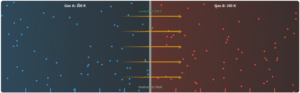
System A Temperature
350K
System B Temperature
250K
System C Temperature
300K
Initial Temperatures
System A (Blue)
350 K
System B (Red)
250 K
System C (Green)
300 K
Simulation Steps
Heat Transfer Rate
Medium